This article was originally published in Garden Culture Magazine, Issue 1 and appeared uner the name, “Carbon Dioxide in Plants and Greenhouses”
There are a couple of things that plants can’t live without. Carbon dioxide is one of these substances. It’s a gas, commonly found in the atmosphere. Although it isn’t as common as oxygen and nitrogen, which make up around 99% of the atmosphere’s volume, carbon dioxide is still relatively common. Over the past decade or so, carbon dioxide has been in the news frequently and has suffered from a bad reputation for being a greenhouse gas that contributes to global climate change. This conclusion has some merit since it is often added to greenhouses to boost plant production. This article is a short introduction into the how and why of carbon dioxide addition in greenhouses.
Plants like all living creatures need energy to survive. Animals get this energy feeding on plants or other creatures. But plants don’t feed on other creatures. Plants store the energy they need by combining elements from soil and air using sunlight to power the reactions. A radically different survival strategy compared to animals. This method of energy harvesting is commonly known as photosynthesis.
In essence, photosynthesis isn’t all that complicated, however when inspected more closely one will find out that it is a cascade of many chemical reactions. To make this a little less complicated photosynthesis can be split up in two parts; ‘Light reactions’ and ‘light-independent reactions’.
LIGHT REACTIONS
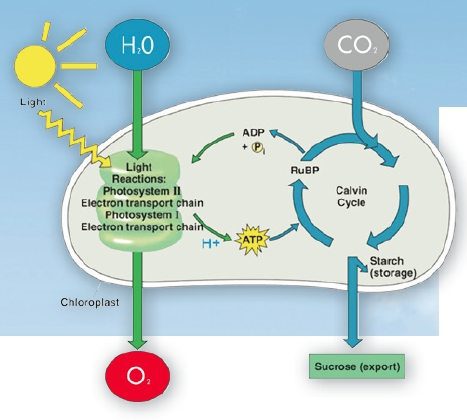 Light reactions are the first part of photosynthesis. These reactions require light in order to work. When a photon from the sun or a grow light strikes the photoreceptive pigment called chlorophyll, water is split into two oxygen molecules and one positively charged hydrogen atom, also known as a proton. These protons, in turn, are used by the plant in the light-independent reactions. But most importantly, the plant uses the energy of the protons to convert ADP (adenine diphosphate) into ATP (adenine triphosphate) by adding a phosphorous group. This is how plants store energy in a usable way, essential in many vital processes. The ATP can be used as a means to transport chemical energy because when it is converted back into ADP energy is released.
Light reactions are the first part of photosynthesis. These reactions require light in order to work. When a photon from the sun or a grow light strikes the photoreceptive pigment called chlorophyll, water is split into two oxygen molecules and one positively charged hydrogen atom, also known as a proton. These protons, in turn, are used by the plant in the light-independent reactions. But most importantly, the plant uses the energy of the protons to convert ADP (adenine diphosphate) into ATP (adenine triphosphate) by adding a phosphorous group. This is how plants store energy in a usable way, essential in many vital processes. The ATP can be used as a means to transport chemical energy because when it is converted back into ADP energy is released.
The molecule can then be recycled into ATP again. The resources the plant has to invest in order to perform these reactions is always the same. However, not all wavelengths of light are equally effective at stimulating chlorophyll, and some wavelengths transfer no energy at all. In general, plants are most efficient in the blue and red ranges of the spectrum. Greenlight is poorly absorbed by chlorophyll. Instead, it is reflected in our eyes, which makes plants look green to us.
LIGHT-INDEPENDENT REACTIONS
The dark reactions then follow up on the light reactions. This is where carbon dioxide comes into play. The proteins that were created during the light reactions are now used to fixate carbon. Carbon fixation is performed by plants using a process called the ‘Calvin Cycle’. A number of different chemicals, including the protons, carbon dioxide, and ATP go into the reaction and in the end yield a simple sugar which in turn can be used by the plant to produce a number of other things including larger sugars and starches. Contrary to the light reactions, these light-independent reactions cost the plant energy.
Like with any chemical reaction or biochemical reaction for that matter, a plant has to have all the ingredients to perform photosynthesis. One of these ingredients is going to be the limiting factor to the reaction. One can understand that if enough water and light are available to plants, CO2 could become a limiting factor. This can certainly be the case since CO2 is present in the atmosphere at levels around 360ppm (parts per million). This is where it starts to get more difficult. Most plants benefit from CO2 addition because the enzyme that transports CO2 inside the plant can get distracted. You see, this enzyme can also bond with oxygen, transporting less carbon dioxide into the plant. This makes the process rather inefficient, as oxygen is available at higher concentrations in the atmosphere. These plants, called C3 plants, benefit greatly from added carbon dioxide.
CARBON DIOXIDE PRODUCTION
In a greenhouse, there are a number of ways to produce carbon dioxide. Piles of composting materials can be used to produce carbon dioxide in a greenhouse, however, this has drawbacks when it comes to pest control. A more modern way to increase the CO2 concentration is by burning propane or some other gas. Gas burners can be linked to controllers and sensors that monitor and adjust the burn rate according to the need of the plants. This is the most common method of CO2 production and, besides recycling CO2 from another source, the least costly. Using pressurized cylinders is too expensive and cumbersome.
There are some drawbacks to burning fossil fuels to produce CO2. With improper combustion, there is a possibility carbon monoxide is formed instead of carbon dioxide. This is a poisonous and potentially deadly gas, odorless and definitely something to watch out for.
Carbon dioxide is something every plant needs and is present in the atmosphere in small concentrations. In situations where it’s preferable to grow crops intensively, CO2 is a welcome addition to boost plant growth. The addition of CO2 increases the rate of photosynthesis that is possible in nearly all plants but other factors change too. In order to get the maximum efficiency out of the added CO2, temperature and humidity have to be pushed to an extreme. In reaction to this the speed of every process in the garden increases. This does sound rather easy however a more extreme climate also increases the chances of pests and other problems one would want to avoid in the crops. Carbon dioxide addition requires a lot of attention to do well, but it can be a valuable tool in increasing yields. It is the next step for gardeners who have mastered their indoor growing environment.
I don’t like it.