The Science Corner will help readers learn about complex topics in the horticultural and hydroponic industries without the confusing jargon and technical terminology. This article will tackle the technical topic of cation and anion exchange capacity. The goal is to break things down for you so you can apply that knowledge and achieve bigger yields, improved quality, and lower costs.
The Cat and the Onion
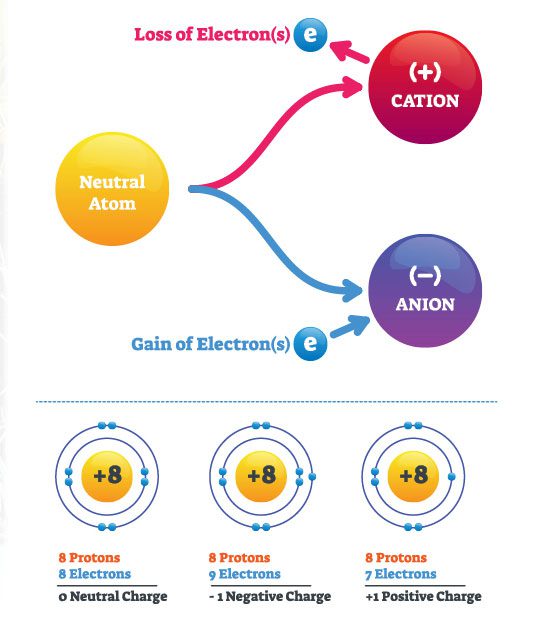
Cations are positively charged particles and anions are negatively charged particles. This concept is essential to understand because all of the nutrients used to grow plants are either positively or negatively charged. If we have a high Cation Exchange Capacity (CEC), the media can hold onto more positive nutrients and deliver them to the plant when it’s needed. Anion exchange capacity is the ability to hold onto negatively charged particles. Still, it isn’t mentioned as often because being positive is much more critical (and the majority of our feed is positively charged).

If you like the simple things in life, know that a low CEC means you have to regularly and consistently add plant food to the media. A high CEC means the media will hold onto excess nutrients and release them when the plant desires.
The way I remember my cations from my anions? Cats are nice (that’s positive) and onions make me cry (that’s a big negative). I told you science corner wasn’t all jargon and technical statements! Just cats and onions.
The Positive And Negative Elements
Plant food is mostly made up of 17 essential plant nutrients:
Macronutrients needed in the highest quantities:
Nitrogen – NO3- Nitrate (Anion)
Ammonium NH4+ (Cation)
Phosphorus – HPO42- (Anion)
Potassium – K+ (Cation)
Micronutrients needed in smaller quantities:
Magnesium – Mg+2 (Cation)
Calcium – Ca+ (Cation)
Sulfur – SO42- (Anion)
Micronutrients needed in trace amounts:
Boron – BO3-3 (Anion)
Chlorine – Cl- (Anion)
Manganese – Mn+2 (Cation)
Iron – Fe+2/Fe+3 (Cation)
Nickel – Ni+2 (Cation)
Copper Cu+/Cu+2 (Cation)
Zinc – Zn+2 (Cation)
Molybdenum – MoO4-2 (Cation)
Non-fertilizer elements obtained through the air and water:
Hydrogen – H+ and H- Carbon, which can form many anions and cations
Oxygen O-2
An easy way of remembering whether an element is a cation or anion is that cations are metallic elements, and anions are non-metallic. Also, the left side of the periodic table will mostly be cations, and the right side is primarily anions.
The Benefits
You may have heard that growing in soil and other complex potting mixes is easier than growing in coco and that growing in coco is easier than growing in Rockwool or water due to the CEC values.
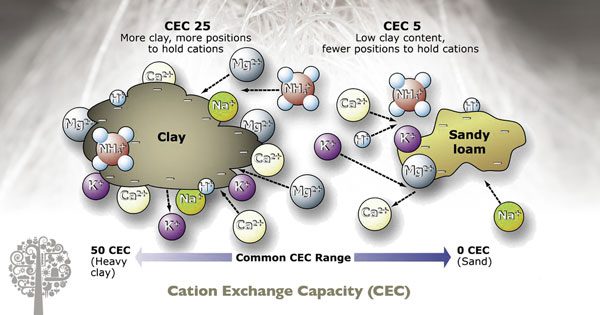
Higher CEC offers room for error without punishment. A substrate with a high CEC will have a lot of clay and organic matter present. They are generally well-buffered, so you will not be affected by large pH swings. On the other hand, low CEC substrates are lightly buffered, if at all, and therefore, can have dramatic fluctuations in pH. For the beginner grower, a soil or high CEC substrate will make life a lot easier and be far more forgiving.
Coco-based substrates are next best as they have good CEC compared to Rockwool or water. Choosing a media with a low CEC comes down to preference. Frequent irrigations and heavy feed schedules don’t work as well with soil or high CEC soils because they tend to retain water for longer, which can decrease root oxygen levels.
Final Message
Cation exchange capacity and positively or negatively charged ions are essential for growth, but they can also be complicated. Understand what CEC is when choosing your media and know that growing with a buffer for potential mistakes will require a high CEC media (such as soil). Frequently irrigated media should have a low CEC.
Lastly, the salt in a substrate has minimal bearing on the quality of the media. Run-off tests are useful to know the EC of a medium during growth or flowering, but provide little benefit with media straight from the bag. Some substrates will be very ‘salty’ (high EC), but this can be due to the levels of calcium, which act as a buffer and increase nutrient uptake. Low EC substrates could retain some of the nutrients you are trying to give to your plant and create deficiencies.
Next, it is crucial to understand how nutrients interact with each other, and the effect of adding too much or too little to the nutrient regime. A great place to start is Mulder’s plant interaction chart, which shows the antagonistic (against) and synergistic (promoting) effects of nutrient interactions. The table makes it clear why nutrients are needed at certain times and why a grower might be experiencing deficiencies or toxicities.
So there you have it; the science of growing easier to understand!
Sources:
Cornell University Cooperative Extension (CUCE) (2007) Cation Exchange Capacity (CEC). Agronomy Fact Sheet Series # 22. Department of Crop and Soil Sciences, College of Agriculture and Life Sciences, Cornell University.
Hazelton PA, Murphy BW (2007) Interpreting Soil Test Results: What Do All The Numbers Mean?. CSIRO Publishing: Melbourne.
McKenzie NJ, Jacquier DJ, Isbell RF, Brown KL (2004) Australian Soils and Landscapes: An Illustrated Compendium. CSIRO Publishing: Collingwood, Victoria.
Moore G, Dolling P, Porter B and Leonard L (1998) Soil Acidity. In Soilguide. A handbook for understanding and managing agricultural soils. (Ed. G Moore) Agriculture Western Australia Bulletin No. 4343.
Rayment GE, Higginson FR (1992) Electrical Conductivity. In ‘Australian Laboratory Handbook of Soil and Water Chemical Methods’ Inkata Press: Melbourne.
Rengasamy P, Churchman GJ (1999) Cation Exchange Capacity, Exchangeable Cations and Sodicity. In Soil Analysis an Interpretation Manual. (Eds KI Peverill, LA Sparrow and DJ Reuter). CSIRO: Melbourne.
Websites visited: